Q1. Inputting API for Wireless Transfer
To use Wireless Data Transfer, an API Key and End user URL must be set in the device.
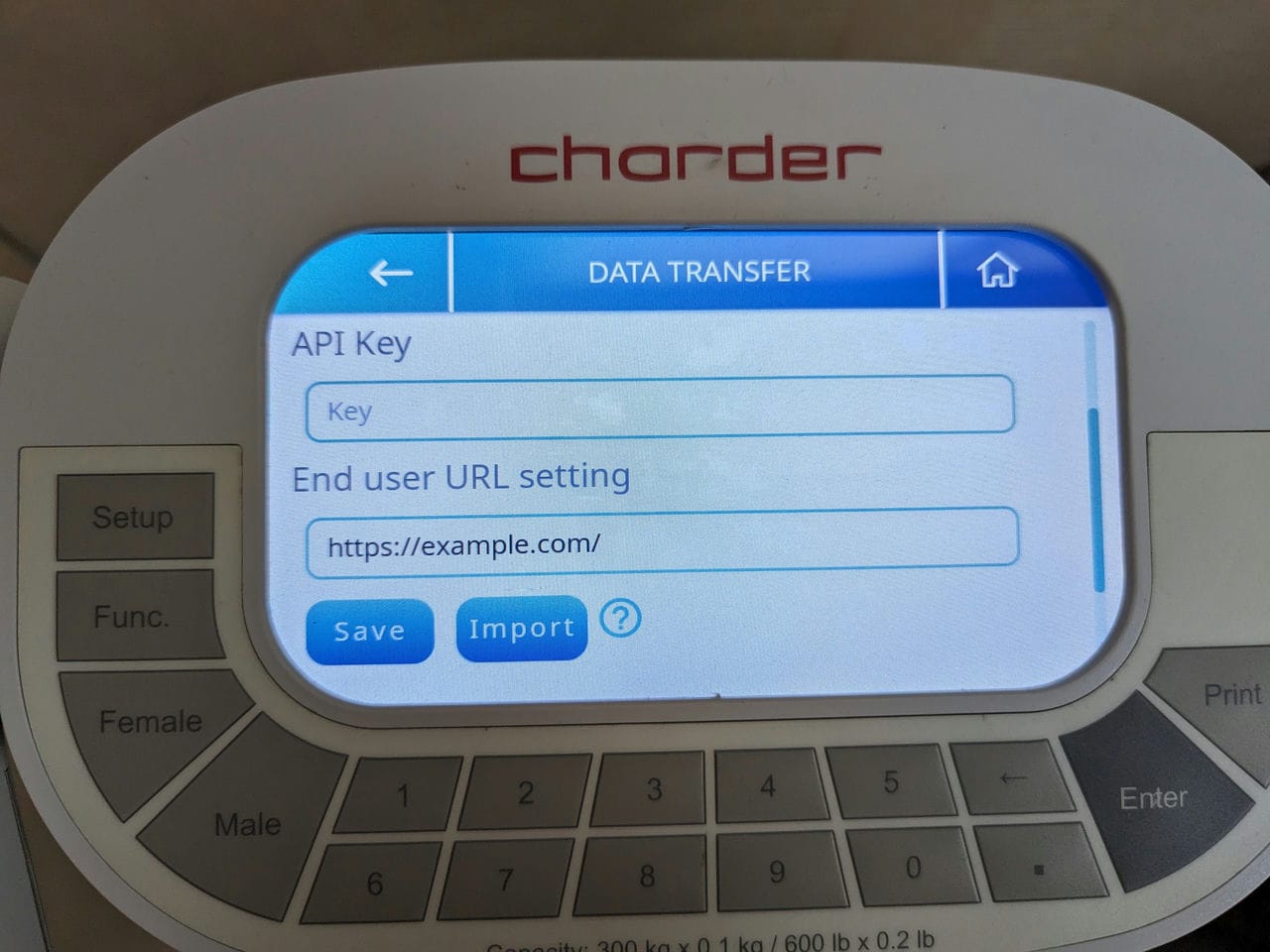
Although the onscreen keyboard can be used, in some cases there may be some special characters in the URL that cannot be input. In addition, it can be inconvenient to type in very long text strings using a touchscreen.
Therefore, the recommended method is to import a properly formatted .txt file to quickly import the API key and URL into the device!
To do so, create a .txt file, with the contents as follows:



Although the onscreen keyboard can be used, in some cases there may be some special characters in the URL that cannot be input. In addition, it can be inconvenient to type in very long text strings using a touchscreen.
Therefore, the recommended method is to import a properly formatted .txt file to quickly import the API key and URL into the device!
To do so, create a .txt file, with the contents as follows:
{
"key":"insert 16-character API key here",
"url":"insert URL here"
}
Save this .txt file into the root folder of a FAT32 USB drive, plug it into the device's USB port(s), enter settings, and select "Data Transfer".
Press "Import", and select the file.
Save this .txt file into the root folder of a FAT32 USB drive, plug it into the device's USB port(s), enter settings, and select "Data Transfer".
Press "Import", and select the file.


Q2. Which devices are compatible with the Charder Insight software?
The MA601 and MA801 are both fully compatible with the Charder Insight PC software. (The U310 is designed for use with Charder ProScan phone/tablet-based software)
Due to continuous updates for devices and software, only certain versions are compatible with one another; please consult the following chart to make sure your versions are compatible, and update if not to avoid compatibility problems!
Due to continuous updates for devices and software, only certain versions are compatible with one another; please consult the following chart to make sure your versions are compatible, and update if not to avoid compatibility problems!
| Device Version | Charder Insight Version |
| v1.0.066 | v1.1.38 |
| v1.0.067 | v1.1.39 |
| v1.0.068 | v1.1.40 |
| v1.0.069 | v1.1.41, v1.1.42 |
| v1.0.072 | v1.1.44, v1.1.45, v1.1.46, v1.1.47 |
| v1.0.077, v1.0.078 | v1.1.51 |
Q3. I'm having trouble importing results
You can export all results for a user using Charder Proscan, and import it into another Charder Proscan app.
(Instructional guide here)
The default file manager used by some Android devices may not support Proscan, and you may need to use a 3rd party file manager to complete the data import action.
(Currently, this issue has been observed in some Samsung devices)
(Instructional guide here)
The default file manager used by some Android devices may not support Proscan, and you may need to use a 3rd party file manager to complete the data import action.
(Currently, this issue has been observed in some Samsung devices)
Q4. Can results by synced with fitness apps?
Charder ProScan results can be shared individually or as a batch to other people using the Charder ProScan app. You can also share results in image format using a wide variety of messaging services.
Currently, Charder ProScan results are not automatically synced with fitness-related apps from other brands (ex: Fitbit/Google Fit/Apple Health).
Currently, Charder ProScan results are not automatically synced with fitness-related apps from other brands (ex: Fitbit/Google Fit/Apple Health).
Q5. What is the difference between "Admin" and "User" accounts?
The "Administrator" account is not used for conducting measurement, but rather for managing user data. You can think of it like a fitness coach's account in a gym, who assists members in conducting measurements. If you are using Charder ProScan as an individual user, we recommend going to Settings→App Settings→Auto login→and select "On" so you don't have to worry about inputting the Administrator ID and Password every single time you want to use the app.
The "User" account is for people measuring themselves using the App; each person will have their own individual account.
The "User" account is for people measuring themselves using the App; each person will have their own individual account.
Q6. How do I share/import measurement data?
Please consult this guide explaining how to use Charder ProScan to share and import measurement results!
Q7. How is "Ideal Weight" calculated?
The "Ideal Weight" range provided by the Charder ProScan app is based on BMI
Q8. Can patients outside the recommended age range use BIA devices?
Charder's Body Composition Analyzers have a specified age range for measurement accuracy. For example, the recommended age range for best results is between 6 and 85 years old for the MA601 and MA801. This is not due to safety concerns - BIA is safe for all ages, and you can safely measure someone that is outside of the recommended age range with no problem!
Body Composition results need to be validated against gold standards to evaluate accuracy, and improve continuously algorithms. Research ethical boards are understandably averse to approving research that may involve radiation (in the case of DXA) and other risk factors for children, especially those under 6 years old. What's more, a 3-year old may be unable to even reach the hand electrodes, or stand on the device long enough to conduct measurements for data collection. As such, while children under 6 can use Charder'sdevices safely, it is hard to prove with empirical data that the same level of accuracy can be guaranteed for a 2-year old, compared to a 6-year old, or other results within the recommended age range.
The same applies to those over 85 years old - while there are no safety concerns, gathering sufficient measurement data for healthy adults over this age can be difficult, especially for the purposes of data validation, where enough males and females within an age bracket, of various different ethnicities are required. Charder expands the age range only after we have sufficient data, ensuring that we stand by the results provided by our devices.
Q9. Can BIA be used by users with stents?
Patients with embedded electronical medical devices (ex: pacemakers) are prohibited from using Bioelectrical Impedance Analysis (BIA) devices, because of the fear of the electrical current used for measurement affecting the embedded device.
Because stents are not powered by electricity, there is no potential safety risk caused by BIA measurement.
Because stents are not powered by electricity, there is no potential safety risk caused by BIA measurement.
In general, there is the potential for slight measurement inaccuracy if the patient has significant amounts of metal embedded in their body, as metal is highly conductive, which may cause the device to misinterpret results. However, stents are not nearly large enough to make a noticeable difference like a large metal implant in your foot might.
Q10. Do more frequencies mean the device is more accurate?
Not necessarily. It is important for the device to have enough frequencies to calculate body composition results. Generally speaking, 250kHz is recognized to be necessary for reliable ICW:ECW calculations. But beyond that, impedance results might be measured but not used.
Q11. What is the difference between a multi-segmental vs a traditional BIA device?
Hand-to-hand BIA devices measure impedance passed between the arms, and the lower body is estimated based on the impedance measured in the upper body. Conversely, leg-to-leg devices measure impedance passed between the legs, and the upper body is estimated. For subjects with balanced upper and lower bodies, these traditional devices may provide reasonably accurate estimates for whole-body values. However, measurement accuracy is compromised if there is significant upper-lower imbalance (ex: due to top-heavy strength training). More importantly, multi-segment technology allows Charder devices to provide calculations for each segment separately, which is absolutely essential for advanced training and tracking.
Q12. How will results be affected by measurement after exercise?
Exercise can cause a temporary change in impedance, and thus may influence measurement results. Intensive exercise can increase blood circulation that may continue for an extended period of times, and we recommend avoiding measurements shortly after exercise.